- Blog
- Meal planner free printable
- Nsfw pokemon discord
- Family halloween movies on netflix 2017
- Floor tileset rpg maker vx ace
- Nexomon extinction all legendary locations
- Purpose of genograms in social work
- Synergy halth and wellness
- Kies for samsung galaxy tab 3 7-0
- Normal ofa hip xray
- Color blind test for kids blue
- What is scid personality test
- SCID symptoms what is ada gene therapy
- Family movies on netflix september 2018
- Youtube convert mp3
- Tuneskit license account
- Gta 5 for pc free download softonic
- Cool terraria house designscom
- Android windows wifi file transfer
- Decibel scale examples
- English script font free
In the absence of these options, one can opt for a transplant from a compatible or partially compatible donor, with a potentially greater risk of incurring graft-versus-host reaction (GvHD), infections and other complications, even fatal in certain cases.įinally, enzyme replacement therapy is available, i.e. When this type of therapy is not feasible, gene therapy represents a therapeutic option: it is based on a single administration of stem cells with the correct gene, which are taken from the patient's own bone marrow, significantly reducing possible transplant reactions against the host. The first choice is represented by the transplant of hematopoietic stem cells from a compatible family donor, which can cure the disease but is available in less than 20% of cases. Today, there are several treatment options for ADA-SCID. – declares Alessandro Aiuti, Professor of Peediatrics and deputy director and head of clinical research of the Institute.
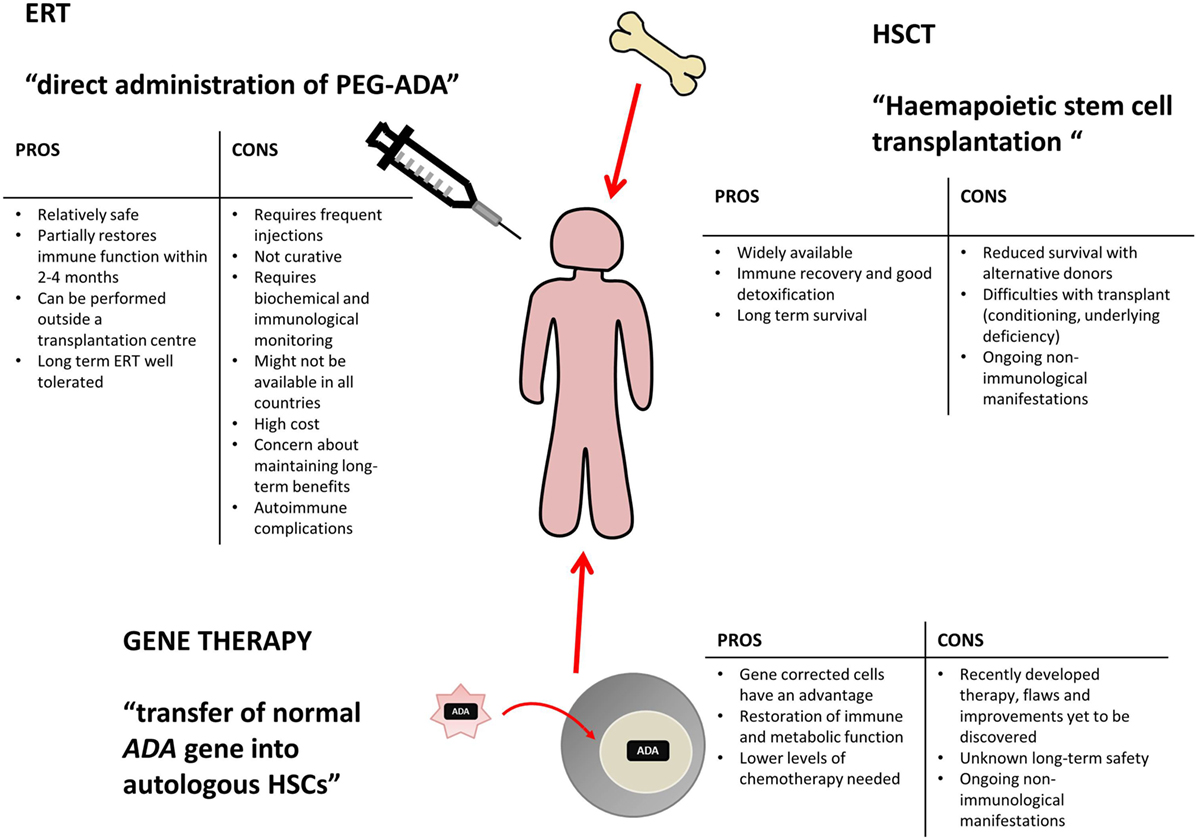
In light of the benefits that we have seen in our young patients in recent years, who are now growing into adults, we can only rejoice in the fact that this gene therapy will continue to be available" A result that is the fruit of the extraordinary work of the researchers and clinicians of the institute, as well as the support of the Telethon Foundation and all the donors. “The research that allowed us to develop this gene therapy began over 25 years ago. The annual incidence is estimated between 1/375,000 and 1/660,000 live births based on data on newborns in the European Union (around 4 million per year), it is estimated that between 6 and 11 children are born with this pathology every year in the 27 EU countries. Children born with ADA-SCID do not have a healthy immune system, so they cannot fight off the most common infections that can be fatal, and are forced to live in a sterile and isolated environment. To date, a total of 45 patients have been treated at SR-TIGET, coming from over 20 countries around the world.ĪDA-SCID (adenosine deaminase deficiency severe combined immunodeficiency), also known as “bubble baby” disease, is a very rare, potentially fatal genetic disorder in which a defective gene blocks the production of an essential enzyme called adenosine deaminase (ADA), necessary for the production and maturation of lymphocytes, a particular type of white blood cell.

This medicine, for which AIFA authorized reimbursement in 2016, is administered at the San Raffaele Hospital in Milan, the only center authorized to date, and is entirely the result of research carried out by the San Raffaele-Telethon Institute for gene therapy (SR-TIGET) and the Telethon Foundation's commitment to transforming these results into a medicine. And today, after the positive opinion of the EMA, the European Medicines Agency, the European Commission approved the transfer of the marketing authorization in Europe for the same therapy to Telethon.

Last year the Anglo-American pharmaceutical company Orchard Therapeutics PLC, owner of the product, announced its intention to disinvest in the field of primary immunodeficiencies. This is Fondazione Telethon's new challenge: for the first time in the world a non-profit organization will assume responsibility for the production and distribution of a medicine for a rare disease, gene therapy for the immunodeficiency ADA-SCID ( Strimvelis). The drug is entirely the result of research by the San Raffaele-Telethon Institute for Gene Therapy (SR-TIGET) in Milan and the San Raffaele Hospital is the only center authorized for its administration After the positive opinion of the EMA, the European Commission approves the transfer of the marketing authorization for the gene therapy for the rare immunodeficiency ADA-SCID from the company Orchard Therapeutics to Fondazione Telethon.

